Are Catalytic Converters Made With Diamond? Essential Truths
No, catalytic converters are NOT made with diamonds. While they contain precious metals like platinum, palladium, and rhodium that are valuable, diamonds have no role in their function. Learn the real materials and why this myth persists.
Hey there, fellow car enthusiasts! Monowar Shohag here, your go-to guide for all things exhaust. Ever wondered what’s packed inside that bulky metal box under your car, the catalytic converter? It’s a common thought, especially when you hear about how valuable they are.
Some folks even whisper that they’re made with diamonds! It’s a fascinating idea, but is it true? Don’t worry, understanding your car’s exhaust system doesn’t have to be complicated. We’ll break down exactly what’s inside your catalytic converter, why it’s so important, and clear up this diamond mystery once and for all. Get ready to discover the essential truths!
The Diamond Myth: Where Did It Come From?
It’s easy to get ideas mixed up in the automotive world. The idea that catalytic converters are made with diamonds is one of those persistent myths. Why does this one stick around? It likely stems from a few places. Both diamonds and the real precious metals inside catalytic converters are incredibly valuable.
People hear “precious metals” and “valuable” and might connect it to other valuable, rare materials like diamonds. Also, the complex internal structure of a catalytic converter, with its honeycomb design coated in chemicals, might look like something high-tech and exotic, leading to wild speculation. But let’s get down to brass tacks—or rather, platinum and palladium!
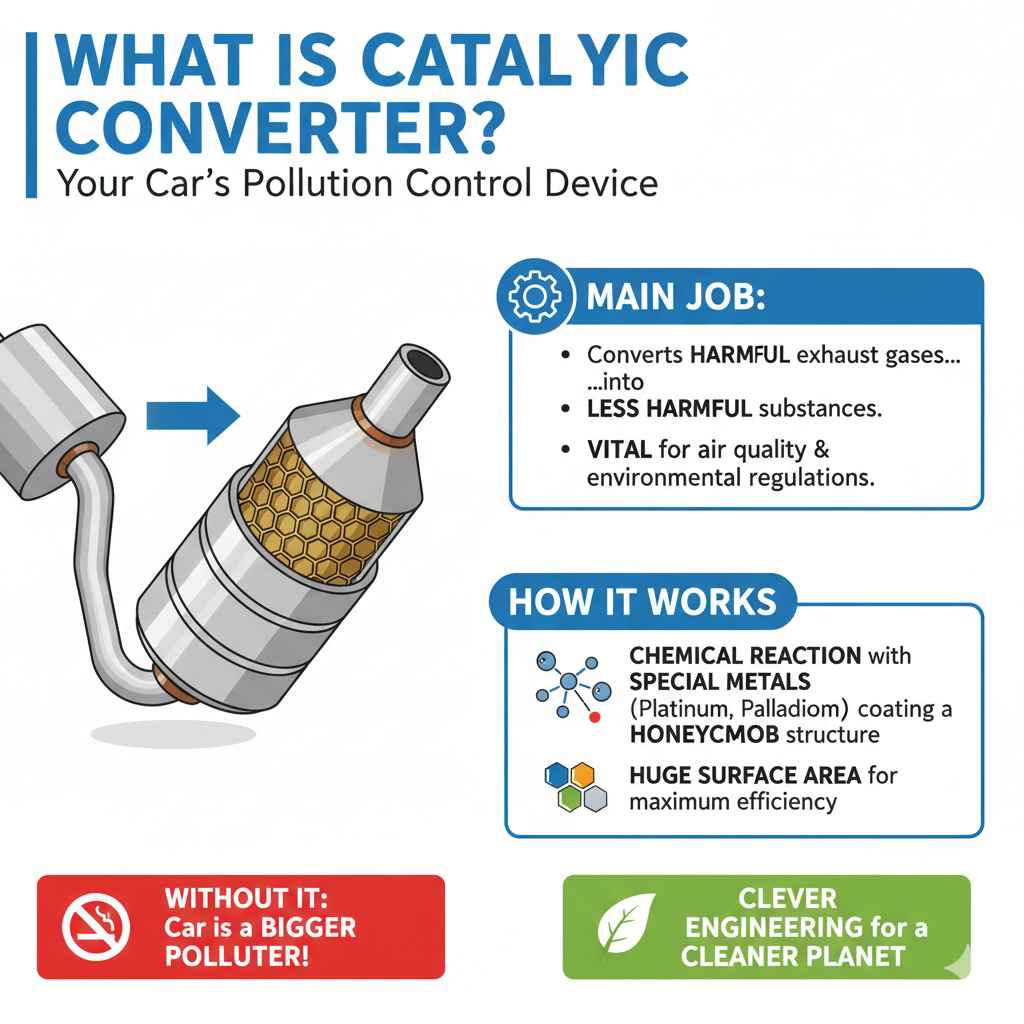
What Exactly is a Catalytic Converter?
Before we dive into what isn’t in them, let’s understand what a catalytic converter is and why it’s a crucial part of your car. Think of it as your car’s personal pollution control device. Its main job is to convert harmful exhaust gases produced by your engine into less harmful substances before they’re released into the atmosphere. This is vital for air quality and meeting environmental regulations. Without it, your car would be a much bigger polluter!
The process involves a chemical reaction, a transformation that happens thanks to special metals coating a honeycomb structure inside the converter. This honeycomb design provides a huge surface area for the exhaust gases to interact with these metals. It’s a clever piece of engineering designed for maximum efficiency.
The Real Precious Metals: Platinum, Palladium, and Rhodium
So, if not diamonds, what gives catalytic converters their value and their amazing ability to clean emissions? The answer lies in three incredibly important and valuable precious metals:
- Platinum (Pt): This is one of the most historically significant metals used in catalytic converters. It’s excellent at helping to oxidize harmful gases like carbon monoxide (CO) and unburnt hydrocarbons (HC).
- Palladium (Pd): In recent years, palladium has become increasingly important and often used alongside or even instead of platinum. It’s particularly effective at reducing nitrogen oxides (NOx)—a major pollutant that contributes to smog.
- Rhodium (Rh): This metal is the superstar for reducing nitrogen oxides (NOx). It works in tandem with platinum and palladium to ensure all major harmful components of exhaust gas are treated.
These metals are used in very small amounts, but their effectiveness is immense. They act as catalysts, meaning they speed up a chemical reaction without being used up themselves. This is why they are so critical to the converter’s function.
Why Are These Metals So Valuable?
The value of platinum, palladium, and rhodium comes from their rarity, their unique chemical properties, and their widespread industrial use, especially in emissions control systems. The automotive industry is a massive consumer of these metals because, by law, all gasoline-powered vehicles must have catalytic converters to reduce pollution. This high demand, coupled with limited supply, drives up their prices. Major news outlets often report on the fluctuating prices of these precious metals, which can contribute to the public perception of catalytic converters as highly valuable items.
For a deeper understanding of these invaluable elements and their role in industry, you can explore resources from institutions like the U.S. Geological Survey (USGS), which provides extensive data on mineral commodities.
The Structure of a Catalytic Converter
To help you visualize where these metals work their magic, let’s look at the typical construction of a catalytic converter. It’s not just a solid chunk of metal; it’s a carefully designed system:
| Component | Description | Function |
|---|---|---|
| Outer Shell | Typically made of stainless steel, it’s the protective housing. | Provides structural integrity and protection from heat and impact. |
| Ceramic Honeycomb (Substrate) | A ceramic material (like cordierite) formed into a dense network of tiny channels or cells. | Offers a massive surface area for the catalytic coatings, efficiently exposing exhaust gases to the active metals. This design maximizes the chemical reaction space. |
| Washcoat | A porous layer, often made of aluminum oxide, applied to the ceramic honeycomb. | Further increases the effective surface area and provides a stable base for the precious metals. |
| Precious Metal Catalyst Layer | Extremely thin layers of platinum, palladium, and rhodium dispersed onto the washcoat. | The active sites where the chemical reactions occur to convert pollutants. |
| Inlet and Outlet Pipes | Connect the converter to the exhaust manifold and the rest of the exhaust system. | Allow exhaust gases to enter and exit the converter. |
As you can see, it’s a sophisticated assembly. The honeycomb substrate is essential, and the precious metals are the hardworking agents of change. Diamonds, with their extreme hardness and inertness, would simply not be suitable for facilitating these specific chemical reactions. They don’t have the necessary catalytic properties, and their structure isn’t designed for this application.
How a Catalytic Converter Works: The Three-Way Process
Modern catalytic converters are often called “three-way” converters because they tackle three main types of harmful emissions simultaneously:
- Reduction of Nitrogen Oxides (NOx): Rhodium is the key player here. It strips oxygen atoms from NOx molecules, converting them into harmless nitrogen gas (N₂) and releasing it into the atmosphere.
- Oxidation of Carbon Monoxide (CO): Platinum and palladium help here. They react with carbon monoxide and oxygen from the exhaust to form carbon dioxide (CO₂), a less toxic gas.
- Oxidation of Unburnt Hydrocarbons (HC): Platinum and palladium also catalyze reactions between unburnt fuel (hydrocarbons) and oxygen, turning them into carbon dioxide (CO₂) and water (H₂O).
For these reactions to happen efficiently, the catalytic converter needs to reach a high operating temperature, usually between 400°C (752°F) and 800°C (1472°F). This is why they are located relatively close to the engine in the exhaust system.
Why Diamond is NOT Used in Catalytic Converters
Let’s address the diamond question directly because it’s important to be clear. Diamonds are made of pure carbon arranged in a crystal structure. They are famous for their incredible hardness and brilliance. While diamonds are valuable and rare, their physical and chemical properties make them unsuitable for catalytic converters:
- Inertness: Diamonds are very unreactive. For a catalytic converter to work, the materials involved must be able to participate in chemical reactions to break down and reform pollutants. Diamonds are too inert for this task.
- Lack of Catalytic Properties: Platinum, palladium, and rhodium have specific electron configurations that allow them to bind with pollutant molecules temporarily, facilitating their conversion. Diamonds do not possess these catalytic properties.
- Thermal Stability vs. Reactivity: While diamonds can withstand high temperatures, they don’t facilitate the necessary chemical interactions at those temperatures. The precious metals are chosen precisely for their ability to be active catalysts within the operating temperature range of an exhaust system.
- Cost-Effectiveness for Function: Even if diamonds could somehow be made to work (which they can’t), their use would be astronomically expensive and impractical compared to the highly effective precious metal catalysts currently employed.
The automotive industry relies on well-established scientific principles and materials that have proven effectiveness. The precious metals used are selected for their specific catalytic capabilities, a role diamonds cannot fill.
DIY Checks and Maintenance (Safety First!)
While you won’t be dealing with cutting or polishing diamonds, there are some basic checks you can perform related to your catalytic converter to keep your car running smoothly and legally. Remember, safety is paramount when working on any part of your car, especially the exhaust system, which can be very hot.
What to Look For:
- Check Engine Light: This is your car’s way of telling you something is wrong. A P0420 or P0430 code often indicates a catalytic converter issue, but it could also point to problems with oxygen sensors or other exhaust components.
- Reduced Engine Performance: If your catalytic converter becomes clogged, it can restrict exhaust flow. This might feel like your car is sluggish, hesitates when accelerating, or has a significant loss of power.
- Strange Smells: A failing converter might emit a rotten egg smell due to unburned fuel.
- Rattling Noises: The ceramic honeycomb inside can break apart over time. If this happens, you might hear rattling sounds from under the car.
- Visual Inspection (from a distance!): After the car has cooled down completely, you can look under your car to check if the catalytic converter casing is rusted, damaged, or shows signs of tampering. However, extensive inspection is best left to professionals.
What NOT to Do:
- Never work on a hot exhaust system. Always let your car cool down completely for several hours.
- Wear protective gear like gloves and eye protection.
- Avoid crawling under the car without proper support, such as jack stands. Never rely on a jack alone.
- Do not attempt to remove or “clean” your catalytic converter yourself unless you have the proper tools, knowledge, and safety equipment. Tampering with emissions control devices can lead to fines.
If you suspect an issue, it’s always best to consult a qualified mechanic. They have the tools and expertise to diagnose problems accurately and safely.
Catalytic Converter Theft: A Disturbing Reality
Unfortunately, the true value of catalytic converters has led to a rise in theft. Because of the precious metals inside, thieves specifically target them. They can be cut off from a vehicle in a matter of minutes, often leaving the owner with a loud, undrivable car and a significant repair bill. This is a serious issue that many car owners face today. Laws and enforcement are trying to combat this, but prevention can be difficult. Some security measures include:
- Parking in well-lit areas.
- Installing protective shields if available for your vehicle model.
- Engraving your Vehicle Identification Number (VIN) onto the converter (check local regulations).
- Being aware of your surroundings.
The theft is a direct consequence of the valuable metals contained within, not because of any diamonds.
Recycling and Environmental Impact
One positive aspect of the precious metals in catalytic converters is their potential for recycling. When a converter reaches the end of its life or is replaced, the valuable platinum, palladium, and rhodium can be extracted and refined. This process is crucial for reducing the need for new mining, which has its own environmental impacts. Companies specialize in recovering these metals, turning old, spent converters into valuable raw materials for new ones. This closed-loop system is an important part of sustainable manufacturing.
For more information on metal recycling, you can check out resources from organizations like the Environmental Protection Agency (EPA), which discusses metal recycling and its benefits.

Catalytic Converter FAQs
Let’s answer some of your most pressing questions about catalytic converters:
Q1: Are catalytic converters always working, or do they need to be replaced?
A1: Catalytic converters are designed to last for many years, often tens of thousands of miles. However, they can fail due to age, overheating, contamination from engine oil or coolant, or physical damage. When they fail, they need to be replaced.
Q2: How much does a catalytic converter replacement cost?
A2: The cost can vary significantly depending on the make and model of your vehicle, as well as the specific converter required. Because of the precious metals inside, new catalytic converters can be expensive, often ranging from several hundred to over two thousand dollars, plus labor.
Q3: Can I drive my car if my catalytic converter is missing or stolen?
A3: You can briefly start and move your car, but it’s not advisable for longer distances. Your car will be extremely loud, poorly performing, and will fail emissions tests. Driving without one can also potentially damage other engine components due to unburnt fuel entering the exhaust system downstream. It’s also illegal in most places.
Q4: What are the signs that my catalytic converter is starting to go bad?
A4: Common signs include a persistent “Check Engine” light, reduced engine power or acceleration, a rotten egg smell in the exhaust, and rattling noises from under the vehicle. If you notice these, it’s best to get it inspected.
Q5: If my catalytic converter is failing, is it because of my driving habits?
A5: Some driving habits can contribute to premature failure. Consistently driving with a misfiring engine or an issues that causes the engine to run too rich (too much fuel, not enough air) overloads the converter with unburnt fuel, causing it to overheat. Regular maintenance, like ensuring your engine is running correctly and that your oxygen sensors are functioning, helps protect the converter.
Q6: Are there any aftermarket catalytic converters that don’t use precious metals?
A6: While some parts marketed as “direct-fit” “universal” or performance catalytic converters exist, they are still legally required to contain precious metals to meet emissions standards. Generic “test pipes” or “de-cat pipes” bypass the converter; these are illegal for use on public roads in most regions and will cause your car to fail emissions tests. True aftermarket catalytic converters meeting EPA standards will contain platinum, palladium, and rhodium.
Q7: Where can I find more information on exhaust system components?
A7: Reliable sources include your vehicle’s owner’s manual, manufacturer websites, reputable automotive repair sites like YourMechanic or HowStuffWorks, and by consulting with certified automotive technicians.
Conclusion
So, there you have it! The mystery of whether catalytic converters are made with diamonds is solved: they are absolutely not. The truth is, they are intricate systems that rely on the unique and valuable properties of precious metals like platinum, palladium, and rhodium to perform their vital function of cleaning our car’s exhaust. These metals aren’t just valuable; they are essential for meeting environmental regulations and keeping our air cleaner.
Understanding what’s inside your catalytic converter isn’t just about debunking myths; it’s about appreciating the technology that keeps our vehicles running cleaner and more efficiently. Whether it’s understanding the importance of maintenance, being aware of potential theft issues, or knowing when a replacement is needed, you’re now better equipped to care for this critical part of your car’s exhaust system. Keep those engines running smoothly and cleanly!







