Are All Resonance Structures Weighted Equally? Uncover Truth
Have you ever wondered if all resonance structures are given equal importance when it comes to understanding a molecule’s true nature? If you’re diving into the world of chemistry, this question might have crossed your mind more than once.
Resonance structures can be tricky, but unlocking their secrets can transform how you perceive chemical bonds and reactions. Imagine if you could grasp how electrons really behave in a molecule. Wouldn’t that give you a new perspective on the elements that make up our world?
In this post, we’ll explore whether all resonance structures are weighted equally, and why this matters to you. Discover how this knowledge could reshape your understanding of chemical stability and reactivity. Prepare to delve into the fascinating details that will keep you on the edge of your seat, eager to unravel the mysteries of molecular behavior. Your journey through the maze of resonance awaits—are you ready to see beyond the surface?

Credit: www.chemistrylearner.com
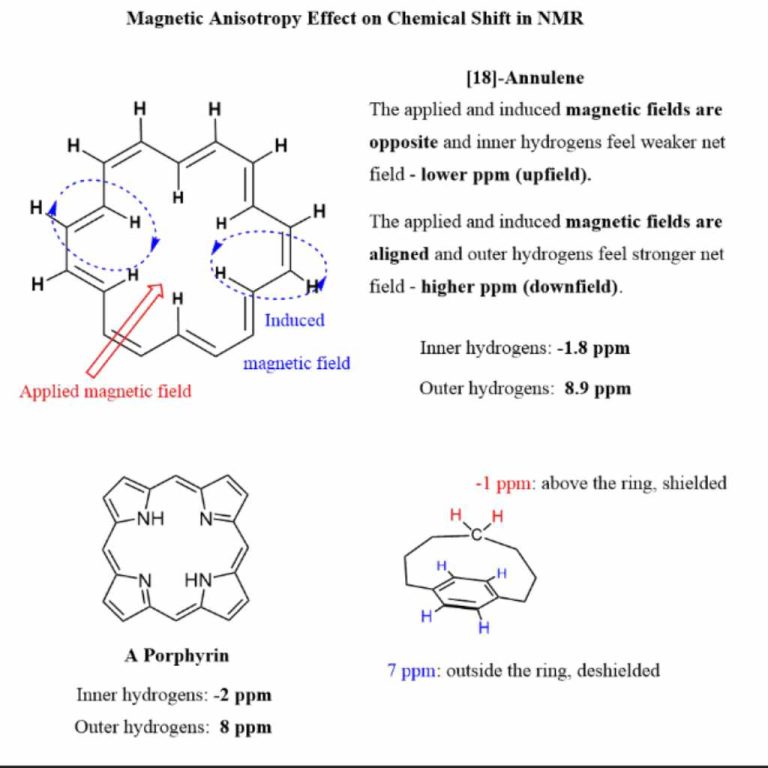
Concept Of Resonance Structures
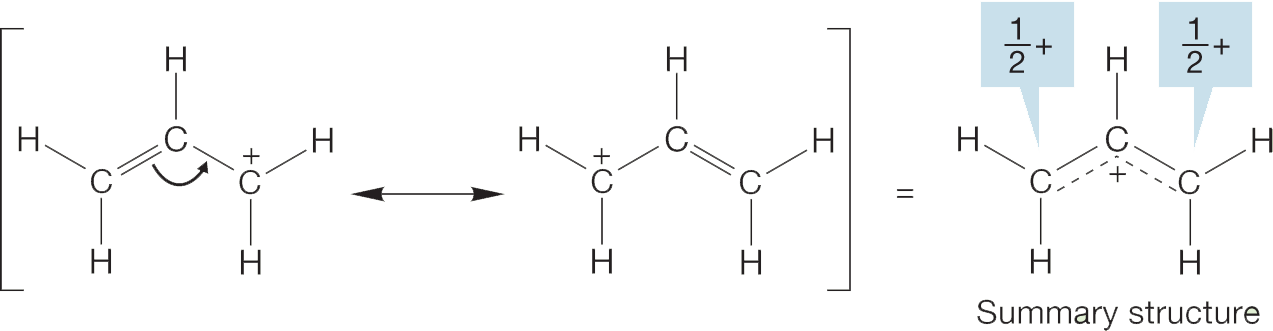
Resonance structures show different ways atoms connect in molecules. These structures do not exist separately. They are a way to explain how electrons move. Electrons can shift between atoms. This helps stabilize molecules. Some structures are more stable than others. Stable structures contribute more to the molecule’s real form. Stability depends on factors like charge distribution and bond strength. Not all resonance structures are equal in importance. Some have higher weight in describing the molecule. Understanding resonance helps in predicting molecule behavior.
Credit: nerd.wwnorton.com
Criteria For Resonance Structure Stability
Charge distribution affects resonance structure stability. Equal charge spread is often better. Uneven charge distribution can make a structure less stable. Charges on atoms should be minimal. Neutral molecules are usually preferred. Negative charges often sit on electronegative atoms.
Resonance structures should follow the octet rule. Atoms should have eight electrons in their outer shell. Stable structures often obey this rule. Incomplete octets can cause instability. Complete octets add to resonance stability. Hydrogen, though, only needs two electrons.
Electronegativity plays a role in resonance. More electronegative atoms hold charges better. Less electronegative atoms struggle with charges. Stable structures align charges with electronegativity. Electrons favor electronegative atoms.
Energy Levels In Resonance
Resonance structures have different energy levelsmolecule behavior. Stable structures contribute more to the real structure.
Bonds can be strong or weak. Strong bonds are shorter. Weak bonds are longer. Resonance affects bond strength. It changes bond length too. Short bonds are usually more stable. Long bonds are less stable. Resonance helps balance these differences. It makes molecules more stable overall.
Influence Of Molecular Geometry
Molecular geometry affects resonance structures. The spatial arrangement of atoms matters. It can change the stability of structures. Some arrangements are more stable. These stable forms influence electron distribution. They often have more weight. Less stable forms have less impact. Geometry helps in understanding these differences.
Hybridization affects the shape of molecules. It changes bond angles. This can alter resonance structure weights. Sp, sp2, and sp3 hybridizations create different shapes. These shapes affect how electrons are shared. The sharing impacts the stability of the structure. Stable structures often contribute more. Unstable ones contribute less. Hybridization plays a key role in this process.
Experimental Evidence And Computational Methods
Spectroscopy helps us see molecules in detail. It shows how resonance structures look in real life. Scientists use this tool to study molecules. Different light helps in seeing different parts. This method tells us about bond lengths and angles. Molecules can change shapes. This change affects resonance structures. Not all structures are the same. Some are stronger or weaker. Spectroscopy gives us clues. This helps in understanding molecules better.
Quantum chemistry uses math to study molecules. It helps predict resonance behavior. Computers solve complex math problems. These problems show how electrons move. Quantum methods show the importance of each structure. Some structures are more important. Others less so. This helps scientists understand molecules. They learn which resonance structures are strongest. Computers make this process easier. They help in predicting results. Quantum chemistry is a key tool in modern science.
Implications In Chemical Reactions
Resonance structures play a big role in chemical reactions. They help decide reaction pathways. One structure might lead to a different path than another. This can change how fast a reaction happens. It might even change what product forms in the end. Some structures are more stable. More stable structures often have more weight. They affect the reaction path more.
Catalysts speed up reactions. Resonance structures can affect how catalysts work. Different structures might change the catalyst’s job. Some structures make catalysts work better. Others might not help the catalyst at all. Knowing which structure is important helps chemists. They can pick the right catalyst for the job. This is vital for making reactions faster and efficient.
Common Misconceptions
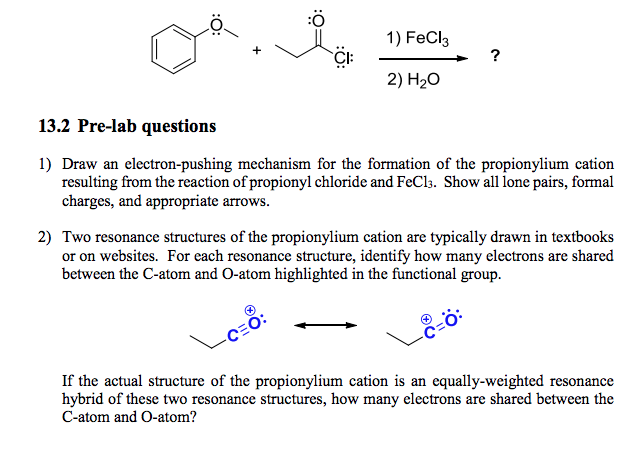
Some believe resonance structures have equal weight. This is not true. Each structure has a different stability. More stable ones have more impact. Less stable ones have less influence. It’s important to recognize these differences.
Models often show resonance as simple. They can be misleading. Simplified models don’t show true complexity. Real molecules behave in complex ways. It’s crucial to understand this. Simplified models can confuse students. They may think all structures are equal.
Credit: www.chegg.com
Frequently Asked Questions
What Are Resonance Structures In Chemistry?
Resonance structures are different Lewis structures for the same molecule. They depict different possible placements of electrons. These structures help visualize electron distribution in molecules. Resonance is important for understanding stability and reactivity. Not all resonance structures contribute equally to the molecule’s actual structure.
Do All Resonance Structures Have Equal Importance?
Not all resonance structures are equally important. Some contribute more to the actual electron distribution. Structures with lower energy and more stable configurations are favored. They have greater influence on the molecule’s properties. Resonance hybrid shows the average electron distribution, highlighting more significant structures.
How Are Resonance Structures Evaluated?
Resonance structures are evaluated based on stability and energy. Structures with complete octets and minimal charge separation are preferred. The most stable structures contribute more to the resonance hybrid. Evaluating resonance helps understand molecule’s reactivity and stability.
Why Do Resonance Structures Matter In Chemistry?
Resonance structures matter because they provide insight into electron distribution. They help predict molecule stability and reactivity. Understanding resonance is crucial for predicting chemical behavior. Resonance influences properties like acidity, basicity, and bond length.
Conclusion
Not all resonance structures are weighted equally. Importance depends on stability and contribution. More stable structures contribute more to the molecule’s behavior. Less stable ones play a minor role. Key factors include charge distribution and bond arrangement. Understanding these helps in predicting chemical reactions.
It’s crucial for students and chemists. Practice identifying which structures matter most. It simplifies complex chemistry concepts. Always focus on stability and the rules. This approach makes learning resonance structures easier. Stay curious and keep exploring. Chemistry is fascinating when you grasp these fundamental concepts.